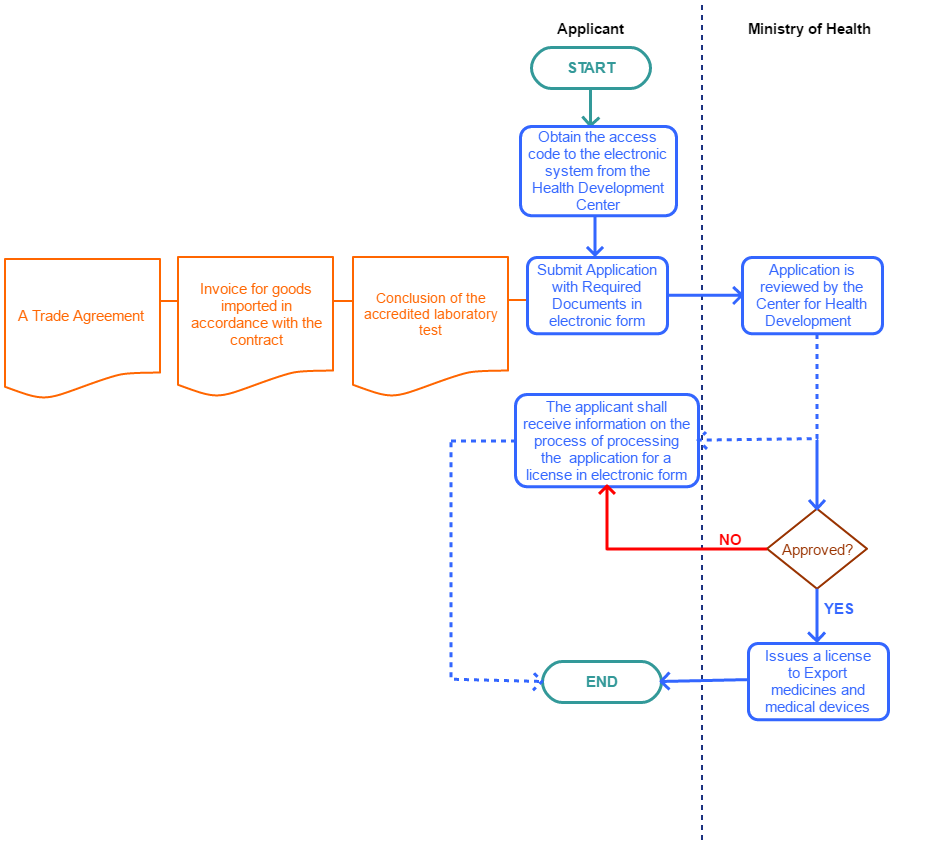
View Procedure
| Procedure Name | Export License issuing procedure for medicines and medical devices | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Description |
Required Documents
Process Steps
NOTE;
| ||||||||||||||||||||||||||||||
| Category | Export |
| Title | Description | Created Date | Updated Date | Issued By |  |
|---|---|---|---|---|---|
| Import/Export License for medicines and medical devices, 2017 Annex 2 to Order A / 407 | This License form applies to traders wishing to import/export of medicines and medical devices. License relates the Import License procedure for medicines and medical devices | 30-07-2020 | 08-10-2020 |
| Name | Measure Type | Agency | Description | Comments | Legal Document | Validity To | Measure Class |
|---|---|---|---|---|---|---|---|
| License requirement for exporting medicines and medical devices | Licensing Requirement | As per the Order on Import and export of medicines and medical devices, this measure applies to traders who want to export medicines and medical devices any other country from Mongolia. | A License applicant shall have a license to engage in professional activities in the field of import and export of medicines and medical devices. The application for a license shall clearly indicate the type, dosage, packaging, name of the manufacturer, quantity and border crossing of the drugs and medical devices to be imported in the shipment. | On Approval of Procedure/Issuing import and export licenses for medicines and medical devices/, 2017 | 31-12-9999 | Good |